Ethylene Vinyl Acetate (EVA) film and Polyvinyl Butyral (PVB) film are two different polymer materials that can be used for glass laminated structures. PVB has been the reference material for the glass lamination on building construction and transportation industries for many years. EVA is challenging current PVB as a laminated material because of some advantageous properties. In fact is a good material also for such type of applications, and is able to fulfill all the key properties that are required to PVB today. Not just that, it is also able to overcome some of the drawbacks of the PVB, outperforming PVB.
Some of the excellent properties of EVA have been already proved in the photovoltaic market. In that market, EVA demonstrated that fact has been already proved by the millions of squared meters of EVA used in photovoltaic. EVA is the “king” material used for the lamination and construction of photovoltaic modules, to produce energy for long periods of time under extreme operation conditions. At this point we should remember that the photovoltaic modules are being produced and sold with 20 year warranty period. Currently other materials have demonstrated to be much more efficient than EVA for such type of applications (eg. Polyolefin (POE)).
 Si lo prefiere, aquí tiene la versión en castellano
Si lo prefiere, aquí tiene la versión en castellano
EVA as well as PVB are thermoplastic polymers with some inherent elastic and natural adhesive properties. Compared with the other ones, rigid thermoplastic materials, at room temperature these ones look as “chewy gummy” products. In that sense, they are different from the rigid form thermoplastics and that is why they are named as “soft” polymer materials.
Although both are thermoplastic materials, they are different, mainly because their chemical structures are different, as you can notice in these figures:
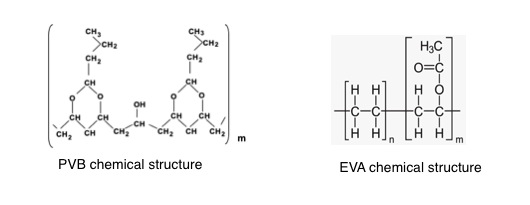
From their chemical structures, we can appreciate that PVB basically has got an only basic unit that repeats m times in the polymer chains (see that there is only one molecular structure between brackets) while EVA has got two (see that there is two different groups of structures together). EVA is composed of two different repetitive units (figure: type units n and type units m), depending on the number (n times in one case and m in another) and depending on how they are distributed into the molecular chains they could give to EVA different properties and performance. According to EVA’s molecular composition we can see different types. For this reason, EVA can be used in an extended range applications because is so much functional thermoplastic than PVB.
Due to their molecular structures and arrangements they are amorphous polymer materials, and that is why both of them have excellent optical properties, and the reason of why they have been used to laminate glasses for Architectural applications.
But it is not just on matters of their optical properties why they can be used in Architectural applications, it is also because of their mechanical properties. Their elastic modulus properties give them the needed properties to dilute the glass-glass dimensional and thermal dilatation tensions when in operation under different weathering environments.
For architectural constructions security is a critical parameter that always should be warranted regardless of the type of glass laminate. That is why, for example to construct balustrade walls, it is not allowed to be built them with just a single piece of glass. Legislations force to use laminated glass systems to insure that when by any reason there is a breakage of one of the glasses, the other one will be able to sustain the small glass pieces adhered to its surface. Safety is first and legislations protect users, by requiring the use of laminated glasses. The process of lamination is looking for implementing of the security element between glasses. The main objectives of the non-glass element is to provide flexibility to the structure an also a good adhesion of the glass in case of glass breakage.
Either the PVB or the EVA are good materials for such a purpose. The differences between them show up in terms of their processability or the final mechanical properties (dilatation coefficients, young modulus, elastic modulus) of the product depending their final application and use. There is where the chemical and physical properties of both materials are important. The above mentioned differences in chemical composition and structure between EVA and PVB allow us the Chemist, Polymer Scientist and Chemical Engineers, play with the raw materials to convert and transform them into specific products with differential and desired performances. Even though it is true that we can manipulate these materials and “drive” them to behave in some extension along our desired performances, for such type of applications where safety is really important, any kind of “gummy materials” we might think to use, they are forced to reach some minimum value properties to fulfill the required security standards.
Having said that, it is important to mention that not all EVA or PVB materials are the same. Under the acronyms of EVA and PVB many different performance products might be considered. It is important to distinguish all of them by looking at their PDS (Product Data Sheets) that manufacturers need to provide. This is an important information to know by the glass lamination companies in order to produce the highest quality lamination glass products according their lamination equipment and processes. But this is not all, at this point here it is important to point out that the relevant properties of the laminated product are the final ones, we mean the ones that any laminate product should fulfill after these materials are laminated and are verified by norms by qualified certification institutes.
Another important property that needs to be fulfilled for this type of products is the adhesion to glass. As it has been previously mentioned, both polymer materials have acceptable intrinsic adhesion properties, but they can always be increased by the use of special additives to the corresponding polymer material. In principle EVA is a better adhesion material than PVB. PVB has an acceptable to low adhesiveness to glass. It has also a high higroscopicity.
In terms of adhesion, besides EVA and PVB, other materials can be used to laminate glasses. But most of them are liquid products that require to mix up at least two different solutions to react. This is not an easy job from the lamination manufacturing point of view. Moreover, as a final product one might end up with a glass adhesion interlayer with different degrees of elasticity that might not fulfil the required mechanical properties needed to be a good a glass interlayer element. The EVA and PVB, intrinsically are being supplied as films, they are easy to handle and to manipulate in the preparation of the glass laminates before they are transformed and integrated as the flexible element of the structured product. The use of films in the lamination process is a great manufacturing advantage compared to the use of liquid materials, even if part of the excellent adhesion is not reached by the use of the films.
One of the main differences between EVA and PVB is their water vapor transmission rate, coming from natural water or weather humidity. The following figure compares the Water Vapor Transmission Rates of the two materials at the same range of temperatures:
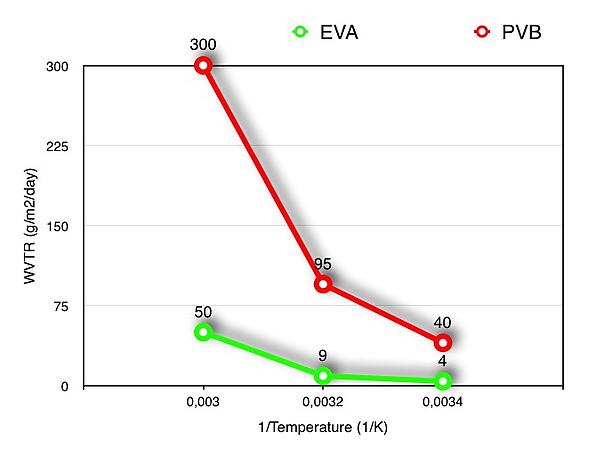
As the graph clearly shows the PVB has an average of 8-9 times higher tendency to permeate water than EVA. This property gives to EVA a great advantage compared to PVB in terms of transportation, storage and use. Not just that also in terms of product resistance against weathering conditions. Normally due to capillarity PVB laminates suffer from water penetration around the edges and glass lamination pieces angles affecting negatively the durability and the mechanical resistance of the laminate. The following picture shows how de-lamination might happen in a floor glass laminated structure that is subjected to periodical cleaning jobs:

Another important difference between these materials is that EVA, in contrast to the PVB, must be converted into a thermoset product during the lamination process. This process transforms the free and “soup of flowing molecules” in the mass into a chemically Crosslink EVA molecule. The lamination process, converts the interlayer material into an elastic, flexible, but at the same time into a structured and not any more temperature melted product. The laminated structure becomes a new stable and elastic interlayer between glasses that provides for the global structure with the required mechanical properties. The following figure represents the process:
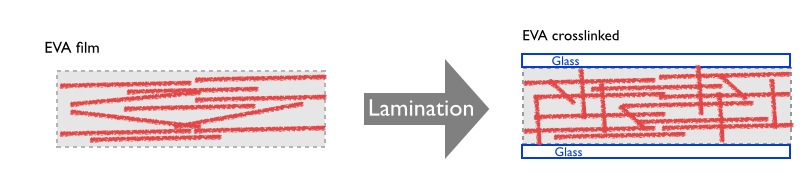
The chemical transformation of EVA might take place in small ovens taking care of vacuum and temperature process control. The cost of the equipment needed is much lower than the one used normally for PVB. The following picture presents one type of IR oven that might be used for lamination processing.

One of the great advantages of EVA is its lower water absorbing compared to the PVB, that allows to store and manipulate the film out of the climatic chambers where PVB needs to be manipulated before lamination.
Moreover, due to the lower adhesiveness properties of PVB this film needs to be processed in autoclaves, where the pressure applied to the lamination structures helps to improve the limited adhesiveness properties of PVB.

In the case of lamination of PVB, there is no chemical cross-linking process between molecules like it happens with EVA lamination processing. The below figure represents the molecular rearrangement of PVB molecules that happens during the PVB transformation process:

If you are interested to get more info, please, just let us know!:
CATEGORIES:
Architectural Glassmarzo 31, 2014
